
Elections & Campaigns, Health Care & Wellness
States Move to Revise Voter-Approved Medicaid Expansion Through Ballot Measures
April 1, 2026 | Mary Kate Barnauskas
February 25, 2026 | Mary Kate Barnauskas

Key Takeaways:
As states consider healthcare affordability issues, Prescription Drug Affordability Boards (PDABs) have emerged as a key debate in state drug pricing policy. Virginia is emerging as a focal point this year, with the Senate passing legislation (VA SB 271) to establish a PDAB with upper payment limit authority, and a similar bill (VA HB 483) under consideration in the House. PDABs are not a new debate in Virginia, as the General Assembly has passed similar legislation twice in the past two years, only for it to be vetoed by former Governor Glenn Youngkin (R). This year, however, the effort has new viability with recently inaugurated Governor Abigail Spanberger (D).
Virginia's debate reflects a broader national trend of states considering PDABs as a tool to address prescription drug costs and affordability.
PDABs are state entities, typically composed of appointed experts in healthcare and the pharmaceutical supply chain, tasked with reviewing drug costs and recommending or implementing cost-containment strategies. PDAB authority ranges by state and can include conducting affordability reviews, issuing reports and recommendations, and setting upper payment limits (UPLs).
Need help tracking state PDAB developments? StateVitals tracks Prescription Drug Affordability Boards and upper payment limit legislation in one place. Explore our PDAB resources on StateVitals →
The most contentious aspect of PDABs has centered on UPLs. In the PDAB process, PDABs conduct "affordability reviews" on select high-cost drugs, and if a drug is determined to pose an affordability challenge, the PDAB may elect to set a UPL. UPLs are limits on the amount that purchasers and payers can be required to pay for a drug when dispensed or administered in the state, and typically apply to all purchases and payments for drugs in the state. UPLs have the potential to significantly alter complex drug reimbursement dynamics and test the boundaries of state authority to regulate drug pricing.
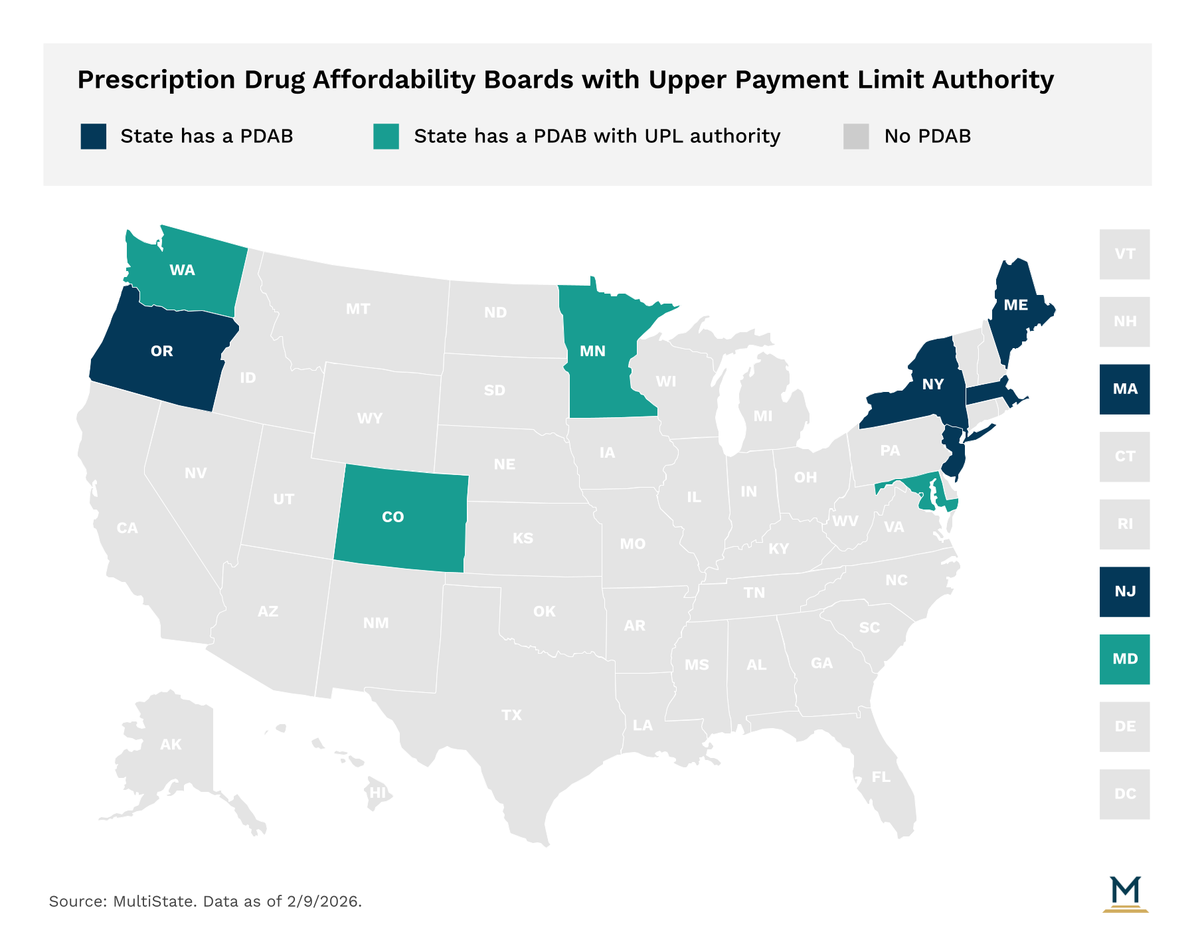
Virginia has emerged as one of the most closely watched states on PDAB legislation in 2026. The General Assembly has now passed PDAB bills in three consecutive sessions — but the first two were vetoed by former Governor Glenn Youngkin (R). This year, with newly inaugurated Governor Abigail Spanberger (D) in office, the legislation has new viability. SB 271, which passed the Senate, and a companion bill, HB 483, which passed the House of Delegates, would establish a Virginia PDAB with upper payment limit authority. Consistent with the trend seen in Colorado, Minnesota, and Maryland, the legislation would require the PDAB to prioritize drugs subject to Medicare negotiation for review and set UPLs at the Medicare maximum fair price.
Whether Governor Spanberger will sign the legislation remains an open question. While prescription drug costs were a prominent part of her campaign, some observers note that Virginia lawmakers have been watching neighboring Maryland (home to one of the oldest PDABs in the country) where relatively little has happened in terms of UPL implementation to date. The outcome of ongoing litigation over Colorado's UPL on Enbrel will also likely factor into how Virginia and other states assess the long-term viability of the PDAB model.
Four states currently have PDABs with UPL authority.
In October of last year, Colorado became the first state to set a UPL on a drug, though it will not go into effect until January 2027. In setting the UPL, the Colorado PDAB relied on the maximum fair price (MFP), the price established through the Medicare negotiation process created under the federal Inflation Reduction Act. The PDAB contended that MFP is a negotiated price that leverages federal research and mechanisms, is significantly lower than other benchmarks, and could lead to savings for payers and consumers.
As other states move towards setting UPLs, MFP will continue to be a key benchmark. In Minnesota, statute requires a UPL to be set at MFP if one exists, and Maryland's PDAB has decided to not set a UPL lower than MFP. In addition, more recently proposed PDAB legislation follows this trend, including the Virginia bill, which prioritizes drugs subject to Medicare negotiation for review. Under the legislation, the PDAB would be required to review such drugs first and set UPLs at MFP.
However, it should be noted that as UPLs move towards implementation, legal challenges have arisen that could shape the future of PDAB and UPL policy. In a closely watched case in Colorado, a drug manufacturer has challenged the state's PDAB over a UPL on its drug, arguing that UPLs conflict with federal authority, attempt to regulate economic activity outside of the state, and violate due process. While a federal district court dismissed the manufacturer's initial challenge when the drug was first selected for a UPL, the manufacturer filed a second lawsuit after the UPL was set, and litigation remains ongoing. As additional states, including Virginia, advance PDAB legislation, the outcome of this case may prove critical in determining whether states can effectively use UPLs as a drug pricing tool.
The ever-evolving state health policy landscape will continue to influence how health care organizations make business decisions. MultiState's team pulls from decades of expertise to help you effectively navigate and engage. MultiState's team understands the issues, knows the key players and organizations. We offer customized strategic solutions to help you develop and execute a proactive multistate agenda focused on your company's goals. Learn more about our Health Care Policy Practice.

April 1, 2026 | Mary Kate Barnauskas

April 1, 2026 | Mary Kate Barnauskas

March 9, 2026 | Lisa Kimbrough, Brock Ingmire